The future of brain tumour liquid biopsies
I caught up with Dr Vineet Datta to discuss the latest breakthrough diagnostic tool
A liquid biopsy is a convenient, non-invasive test that uses a sample of a patient's blood to gather information about the presence and characteristics of cancer. The aim is to detect and analyse circulating tumor cells (CTCs), which have broken away from the primary tumor and are circulating in the bloodstream. Test results may offer a useful tool for early diagnosis and to help track changes in malignancy over time. Real time analysis provides an opportunity to inform treatment decisions and personalise care.
Liquid biopsies for brain cancer have become more reliable over time due to several advancements and improvements in technology and methodology. We now enter a new era, with the world’s first non-invasive liquid biopsy platform to be granted special designation to be used in glial tumours.
Following encouraging results of A prospective, blinded study by a research team at Imperial College London, the USFDA was able to grant Breakthrough Device Designation for Datar Cancer Genetics’ Cancer Detection liquid biopsy solution, '“TriNetraTM-Glio”. The product would primarily be used to help diagnose brain tumours where a conventional biopsy is not possible.
I had the privilege of interviewing Dr Vineet Datta, Executive Director of Datar Cancer Genetics, about the future of this technology and what it means for brain tumour patients.
Andrew: Thank you for your time Dr Datta. Could you please briefly outline your background and how you became involved with this type of research?
Vineet: I have over 20 years of global healthcare experience across clinical patient care, medical assistance, strategic healthcare consulting. My current role as the Executive Director at Datar is to globally expand the adoption of personalised oncology solutions utilising cancer genomics across the clinical mainstream. I have been an active participant in the innovative cancer data research being conducted globally across the Datar platform.
I am a postgraduate in Internal Medicine from JN Medical College, Aligarh, India, and have held various leadership positions across the NHS, International SOS and Apollo Hospitals. I was awarded Diplomas for Membership to the Royal College of Physicians, United Kingdom (MRCP-UK) and the Royal College of Emergency Medicine (MRCEM), London, and am an elected Fellow the Royal College of Physicians and Surgeons, Glasgow, and Fellow, Royal College of Physicians, Edinburgh. I currently also serve as the International Advisor to the Royal College of Physicians and Surgeons of Glasgow, in addition to my professional responsibilities.
Andrew: How would you define what constitutes a ‘liquid biopsy’ in layman’s terms?
Vineet: Genetic tests have diverse purposes, including screening for and diagnosis of genetic disease, the identification of future health risks, prediction of drug responses and identifying targeted therapies to provide customised cancer care. A liquid biopsy is a simple, usually blood-based, non-invasive alternative to surgical biopsies which provides the clinician, a vast range of information about cancer. Traces of the cancer fragments like DNA, RNA and circulating tumour cells (CTCs) are shed in the blood which can also give clues about which treatments are most likely to work for that patient.
Liquid biopsies offer new opportunities to provide insight into the biological and clinical characteristics of a neoplastic disease, and represent alternative routes for diagnostic and prognostic purposes, and for predicting and longitudinally monitoring response to treatment and disease progression. Hence, circulating biomarkers represent promising non-invasive tools in cancer, across the entire ecosystem of disease.

Andrew: What are some of the main challenges remaining considering the applications of this tool?
Vineet: Genomic Medicine is ever changing and reasonably rapidly. Developments in this domain are forcing the clinicians to relook at the way we understand and treat cancer. This dynamic transformation will accelerate, as new technologies become more accessible, easier to use and less expensive. The current challenges include cost, especially self pay models, and the small proportion of patients benefiting from such technologies. While the investment into these technologies is substantial, with higher adoption by clinicians, the cost of these services should reduce, making them more affordable.
The push from various governments on promoting genomics has increased the visibility and adoption of healthcare technologies to improve patient outcomes and services. They need to proactively consider how best to utilise healthcare technologies to benefit a large portion of the population by screening and early cancer detection. Active encouragement of research through collaboration with private healthcare organisations would lead to enhanced applicability of such tools.
Andrew: Developing a reliable and specific liquid biopsy for brain tumours has been notoriously difficult. Many have tried over the years, with varying results. What are some of the obstacles you have encountered and how have you managed to overcome them?
Vineet: Brain tumours and their related cancer biology have always been of active interest to the Datar clinical and scientific teams. Brain cancers represent a serious concern in relation to death due to cancer. Traditionally, imaging and biopsy have been used for diagnosis, however they present challenges with regards to invasive nature of biopsy, hospitalisation and risk of complications. Liquid biopsy assays allow a minimally invasive approach with ease of sampling.
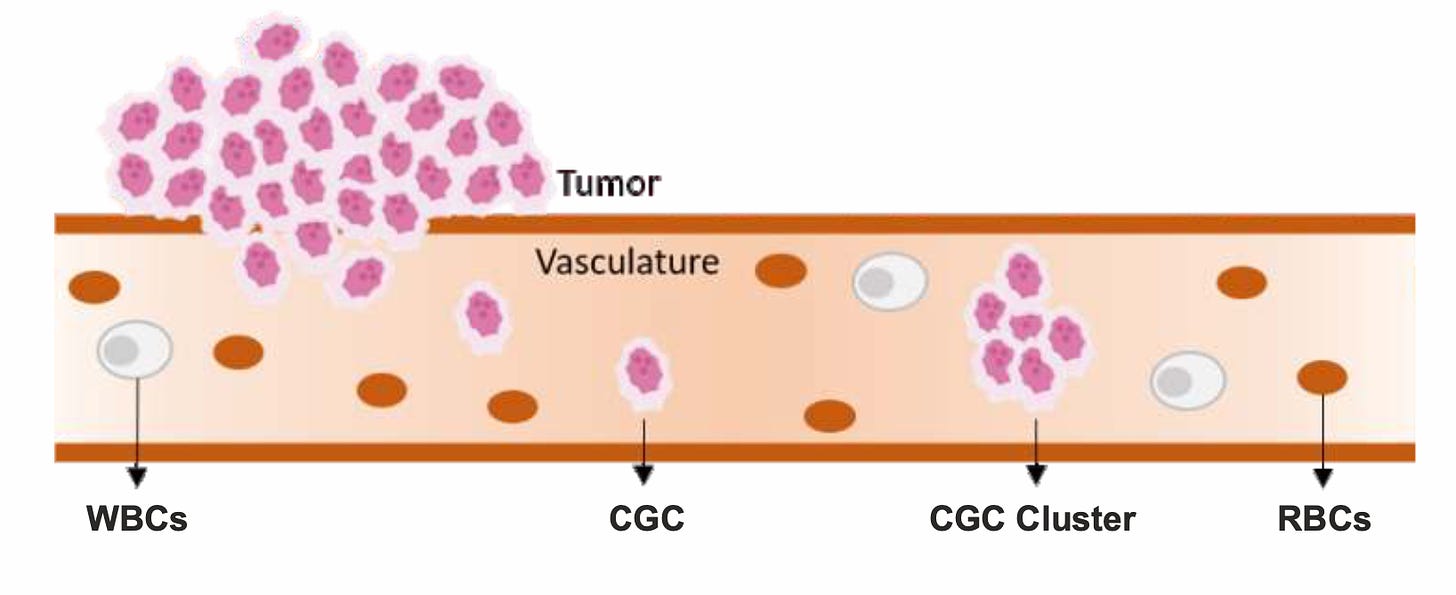
While blood-based liquid biopsies have matured to detect biomarkers in other organ related cancers, progress in the case of brain tumours has largely been limited. One obstacle is the blood-brain barrier (BBB), which hinders efficient passage of cancer biomarkers into the peripheral circulation, for efficient analysis. Presently, no blood test is available for diagnosing brain cancers, and doctors have to rely on complex surgical procedures to obtain tumour tissue for histopathological evaluation.
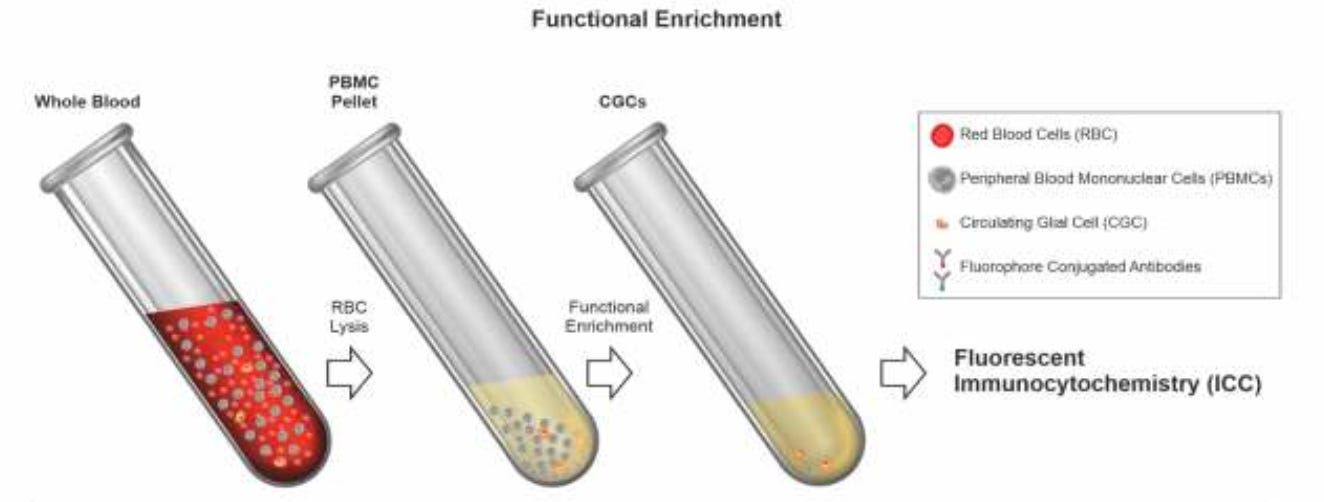
The TriNetra-Glio liquid biopsy is intended to detect the CTCs released in the blood from the brain tumour; these cells are extremely rare and hard to detect. Our novel process for functional enrichment of CTCs is based on a proprietary medium which selectively eliminates all non-cancer cells while permitting malignant cells to survive. This CTC enrichment medium (CEM) is based on the principle that malignant cells have an ability to resist cell death by evading apoptosis. This ability is one of the originally described hallmarks of cancer.
Andrew: Potentially how far in advance could we effectively stratify risk of malignancy?
Vineet: The ability to detect tumour cells and cancer fragments from various cancers, especially the brain, using this methodology represents the breakthrough for inaccessible cancers. Worldwide, brain cancer is the 12th most lethal cancer, and each year, more than 250,000 adults die due to the disease. Diagnosis of brain cancers is resource-intensive, risk-prone and brain biopsies are impossible to perform in up to a third of advanced cases. To aid the neurosurgeon, to provide a liquid biopsy diagnosis from a simple blood test where tumours are deemed inoperable or inaccessible can address an unmet clinical need. There can be limitations of liquid biopsy, in addressing certain therapy related aspects, where tissue samples may be preferred, but stratification of clinical pathways, prior to an invasive, complex procedure, can be made possible utilising such solutions.
Andrew: What types of brain tumour can be detected using the technology and what is the possibility of a false positive result?
Vineet: The TriNetra-Glio liquid biopsy is intended to detect the cells released in the blood from the brain tumour, and is indicated for patients with intracranial space-occupying lesions where a biopsy is unviable due to the inaccessibility of the cancer or a comorbidity/ refusal of the patient, or a non-diagnostic result from a biopsy.
We aim to expand indication for use to include diagnosis of all patients with brain lesions. High sensitivity and specificity was observed in detecting glial malignancies and differentiating them from benign CNS conditions in case controlled studies, and the performance characteristics support its clinical utility for diagnostic triaging of individuals presenting with brain lesions and facilitating more effective diagnosis.
Andrew: How can we best use the information provided from a liquid biopsy with newer imaging tools (such as MR Spectroscopy), possibly even paired with histopathological findings if we can get them?
Vineet: Advances in biomedicine are increasingly able to determine the best treatment approach for some cancers. The shift from a one-size-fits-all approach to tailored offering to individual patients’ needs has begun and is expected to gain greater momentum in coming years.
Evaluation of various technologies is currently ongoing, and while new biotechs continue to provide further insight into the biology of cancer, we will need to evaluate more data on the effective robustness and reproducibility of upcoming clinical information, to determine the utility of such technologies, in isolation or in combination.
Andrew: Given this has received Breakthrough Designation, when can patients realistically expect to have access to the test (either privately, or on the NHS)?
Vineet: The breakthrough designation is a recognition of the potential benefits of our technology in the clinical setting. The test can help individuals where a brain biopsy or surgical resection of the tumour is not possible due to the location of the cancer or other constraints.
The solution has previously received CE certification and is already available to patients privately as 'Trublood-CNS', across the UK and EU regions. The group is actively exploring further regulatory advancements across our services, including possible engagement with public healthcare ecosystems globally, including the NHS, in the near future.
Andrew: What is your vision for the future (short, medium and long term) for this technology and do you think it will become a part of standard screening procedures any time soon?
Vineet: Datar Cancer Genetics is a global oncology research and applications company that is spearheading the development of non-invasive technologies for improved detection, treatment and management of cancer. The group remains committed to expanding its innovative portfolio across the cancer screening and diagnostics ecosystem, advancing our regulatory footprint and solution outreach globally.
The genomic space is rapidly expanding, and I foresee further acceleration across this domain in the near to mid term future, with expanded incorporation of credible and effective technologies into the clinical ecosystem.
Andrew: Your company also offers unique, personalised treatment options. If someone tests positive on one of your cancer tests, what additional/alternative options might you be able to offer to the current standard of care?
Vineet: Datar's state-of-the-art cancer research centre is NABL, ISO, CAP and CLIA accredited.Datar has developed further research and clinical facilities in the UK at Surrey Research Park and the US in North Carolina.
The current Datar genomic portfolio is physician driven, and we work closely with clinical experts who support patients for further testing or referrals across the patient care pathways.
Once again, I must express my gratitude to Dr Datta for his time and thank Datar Cancer Genetics for their work in this area of research.
If you found this article interesting and would like to know more about how to access liquid biopsy tests I would refer you to the Cancer Screening Trust.
The Cancer Screening Trust is a non profit organisation dedicated to providing the latest developments in cancer screening for early detection in over 70 different cancers.