Asking important questions about what we think we know. - Part 1/3
Can new technology and lateral thinking help us to answer some theories we have about cancer metabolism?
I was tempted to title this article - ‘What we think we know about what we know’, but resisted the urge as it could have added some confusion to the attempt at making sense of what is a pretty heavy and curiously complex subject. Maybe I’ve done that anyway, but let’s pretend it never happened and move on.
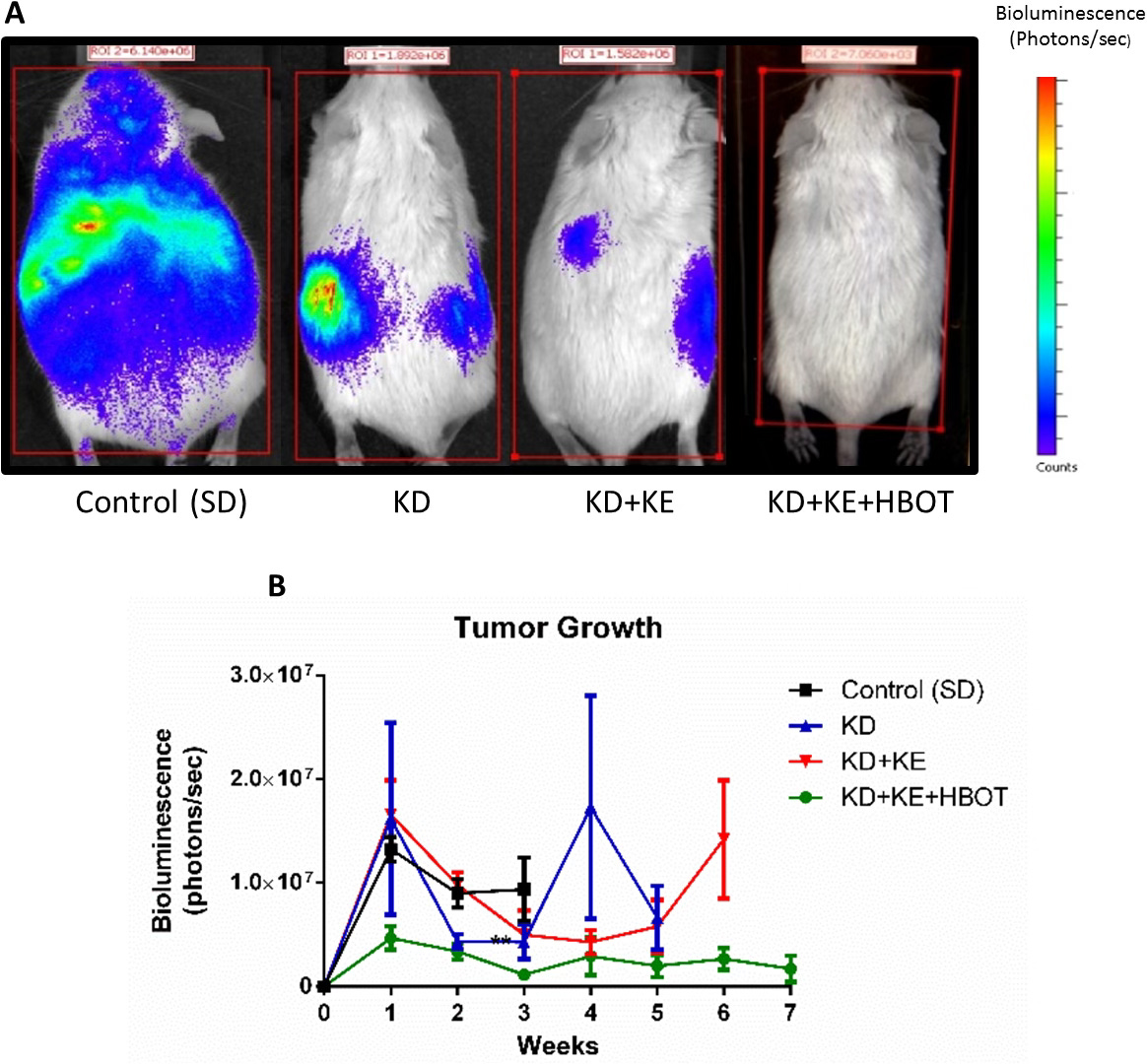
We can go deep into the weeds on this one, but I’ll just touch on some important points. If you’ve been following me for a while, you will know that I view cancer as a metabolic, mitochondrial disease with metabolic solutions. Incidentally, an interesting case study applying ketogenic metabolic therapy (KMT) was recently published on a patient with IDH1 wild type glioblastoma. It illustrates a good example of how to apply the diet therapeutically for cancer.
So far, as a standalone treatment, this kind of approach can only slow down disease progression, unless it is part of a protocol where additional pressure can be applied, placing more stress on cancer cells.
This is why, in my personal case, I took sodium valproate (aka valproic acid, Epilim) at therapeutic doses early on in my cancer journey (due to its anti cancer action in glioma) and adopted a protocol including HBOT, ketone esters (and salts), magnesium in various forms (to manage epilepsy), periodic fasting, boswellia serratta, berberine and cycles of high dose melatonin. Valproic acid is a short chain fatty acid with multiple mechanisms of action against glioma, some of which I have outlined in other articles.
These strategies, are inspired by Professor Thomas Seyfried’s novel ‘Press-Pulse’ strategy to manage cancer, Ben Williams’ story and pre-clinical models of ketogenic metabolic therapies as well as case studies on dogs from Ketopet.

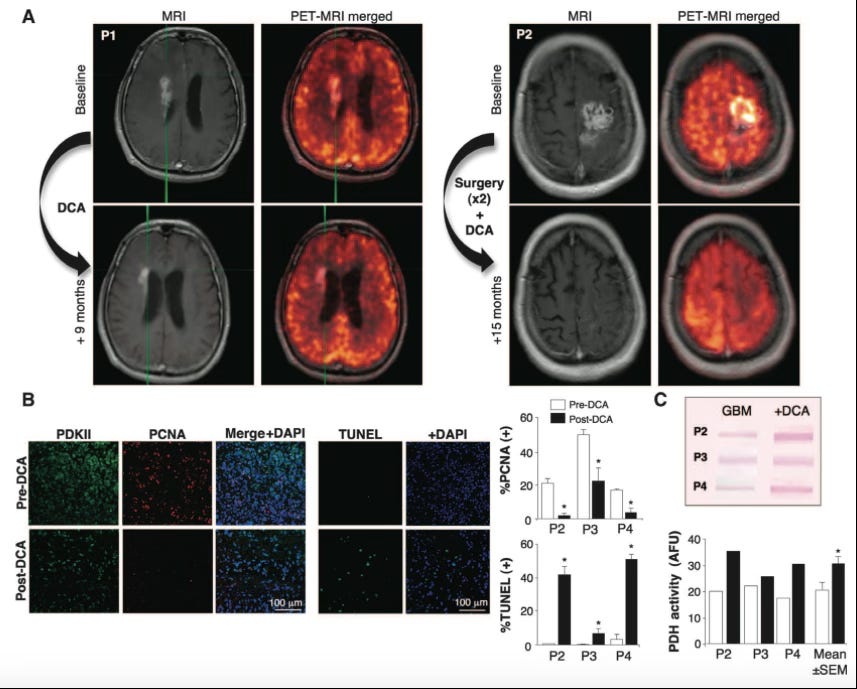
I would have also added Dichloroacetate (DCA) to target the Warburg Effect, however I found I probably wouldn’t metabolise the drug efficiently (see graphic below). Nevertheless, I would add the drug in future should the need arise, being mindful of dose, side effects, etc. and see what happens.
As a side note, it is worth mentioning that when I was on the common anti seizure drug levetiracetam (brand name Keppra), I was supplementing Pyridoxine (vitamin B6), after discovering this is a known deficiency with long term use that can contribute to lethargy and the infamous ‘Kepprage’ - a type of irritability associated with long term use.
My neurologist at the time was clueless about this and other deficiencies caused by these medications, but awareness has since improved. I was on the maximum dose and doing a lot of fasting, so addressing this concern may not be as prescient for others. Also keep in mind that being in a state of ketosis can favourably alter brain chemistry, reducing side effects and increasing the potency of these drugs.

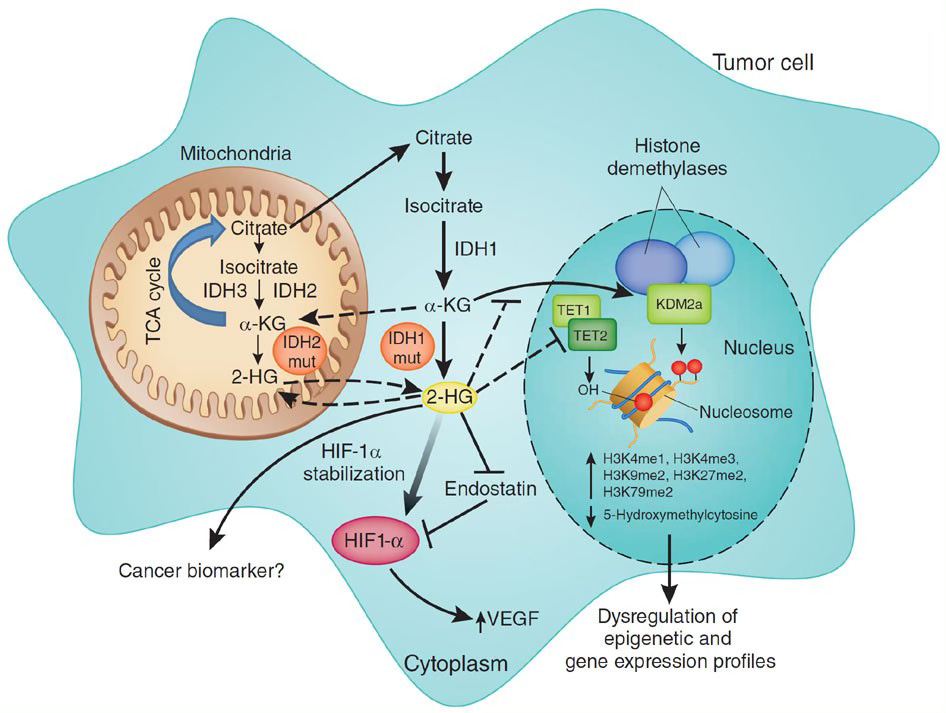
I should have provided more detail about what IDH actually is, but I didn’t want to make your head spin. Even for me this can get ridiculously complicated and I can become lost, but it’s important to clarify.
Isocitrate dehydrogenase (IDH) is an enzyme that catalyses the oxidative decarboxylation of isocitrate, playing a key role in the Krebs cycle. Mutant IDH1 is a gain-of-function mutation that disrupts the conversion of isocitrate to alpha-ketoglutarate (α-KG) and instead favours the production of large amounts of the ’oncometabolite’ D-2-hydroxyglutarate (D-2HG).
Unlike its mutant counterpart, the wild type IDH1 does not produce D-2HG. This means that in cells with normal IDH1, there is no accumulation of D-2HG, avoiding the oncogenic effects associated with this metabolite.
My tumour was IDH1 wild type, which I wasn't too pleased about when I first learned what this could mean. I also had unmethylated MGMT (a DNA repair protein), which is another story, but added to the feeling of uncertainty I had about my prognosis and my reticence to any chemotherapy drugs I might consider.
The IDH1 wild type phenotype is reported to be more aggressive, with a poorer prognosis and resistance to treatment. It is also suggested that this phenotype is more dependent on the amino acid glutamine. But why is this the case, and how much of an issue could it be… if at all?
Metabolic vulnerabilities and IHD1 status
Keep reading with a 7-day free trial
Subscribe to Andrew’s Substack to keep reading this post and get 7 days of free access to the full post archives.


