Deadly brainstem glioma is 'cured' thanks to experimental treatment regime.
Results from the BIOMEDE trial offer hope to children with DIPG brain tumours.
Diffuse Intrinsic Pontine Glioma (DIPG) is a parent’s worst nightmare. This inoperable, paediatric brain tumour is uniformly fatal and devastates families. The reality for children diagnosed with DIPG has been one of little hope. Despite our best efforts, the prognosis remains bleak. Median survival is less than 1 year and for decades now, radiation therapy has been the only real treatment option beyond experimental clinical trials.
The major challenge in treating this type of tumour arises from its location in the pons region of the brainstem, a critical area of the brain. Traditionally, even a biopsy has been considered risky because the pons is responsible for regulating essential bodily functions, including breathing, blood pressure, and heart rate.
In more recent times we have been able to lower the risk of biopsy with stereotactic biopsies, guided by advanced imaging techniques. While this has allowed us to more safely obtain tissue samples for diagnostic purposes, any insights this can provide for treatment remain limited and experimental.
The promise of molecular targeted therapies
Over the past decade, some clues have been revealed about possible treatment targets and who might benefit from targeted therapies, with researchers finding a key genetic mutation, H3K27M, in about 80% of these cases. This mutation affects how genes are turned on and off, a process known as epigenetic targeting. H3K27M, along with other, less common genetic alterations, represents potential targets for new treatments. The success of future treatments will largely depend on biopsies to identify specific molecular characteristics for tailored clinical trial inclusion.
Supported by increasing evidence, there's renewed optimism for advancements in this type of treatment. The BIOMEDE study, with its broad ambition and global collaboration, is pioneering efforts to discover novel treatments for DIPG, previously deemed an 'incurable' cancer.
A common thread in DIPG research, including the BIOMEDE study, is the focus on detailed molecular profiling through biopsies. While this approach is not entirely unique, the study distinguishes itself by prioritising a tailored drug selection strategy for each participant. This personalised approach is essential for customising treatments in clinical trials, ensuring that therapies are more effectively matched to the individual's specific tumour characteristics.
In my opinion this modality offers greater hope to patients than the traditional ‘one size fits all’ approach, which is all too common in the brain tumour world. In the case of DIPG, because there are no other viable options for treatment, and because radiotherapy has the potential to cause great harm to delicate structures in and around this brain region, we are forced into investigating novel therapies.
What makes the BIOMEDE study so special?
The BIOMEDE study has heralded a major breakthrough as it progresses into phase III trials. Having recognised the desperate need to discover novel therapies, this international clinical trial takes advantage of truly ‘personalised medicine’ to find better treatments for DIPG.
In phase II, it compared the effectiveness of three drugs; Erlotinib, Dasatinib, and Everolimus; each chosen based on the results of molecular profiling of biopsied tumour tissue. Spanning 2014 to 2019 and involving 70 centres across Europe, Australia, and New Zealand, the study demonstrated the potential of personalised therapies for DIPG, emphasising the importance of the tumour's biological characteristics.
Everolimus, an mTOR inhibitor, was selected for further investigation due to its favourable safety profile and slightly improved median survival times compared to other drugs, with one patient reportedly achieving complete remission of disease.
The use of Everolimus in this study highlights a strategic shift towards more personalised medicine, with the BIOMEDE study highlighting its potential as part of a novel, highly personalised therapeutic strategy to optimise treatment for DIPG.
The Phase II trial
The phase II trial was completed fairly recently as far as I can gather, with results just published. As part of the inclusion criteria for the trial, patients had not received any other treatment, including radiotherapy.
In the study, all treatments were personalised. Specific drugs were given to each patient, chosen based on the molecular characteristics of their tumour identified through a biopsy.
Below is a graphic illustrating how treatments were decided for each patient:
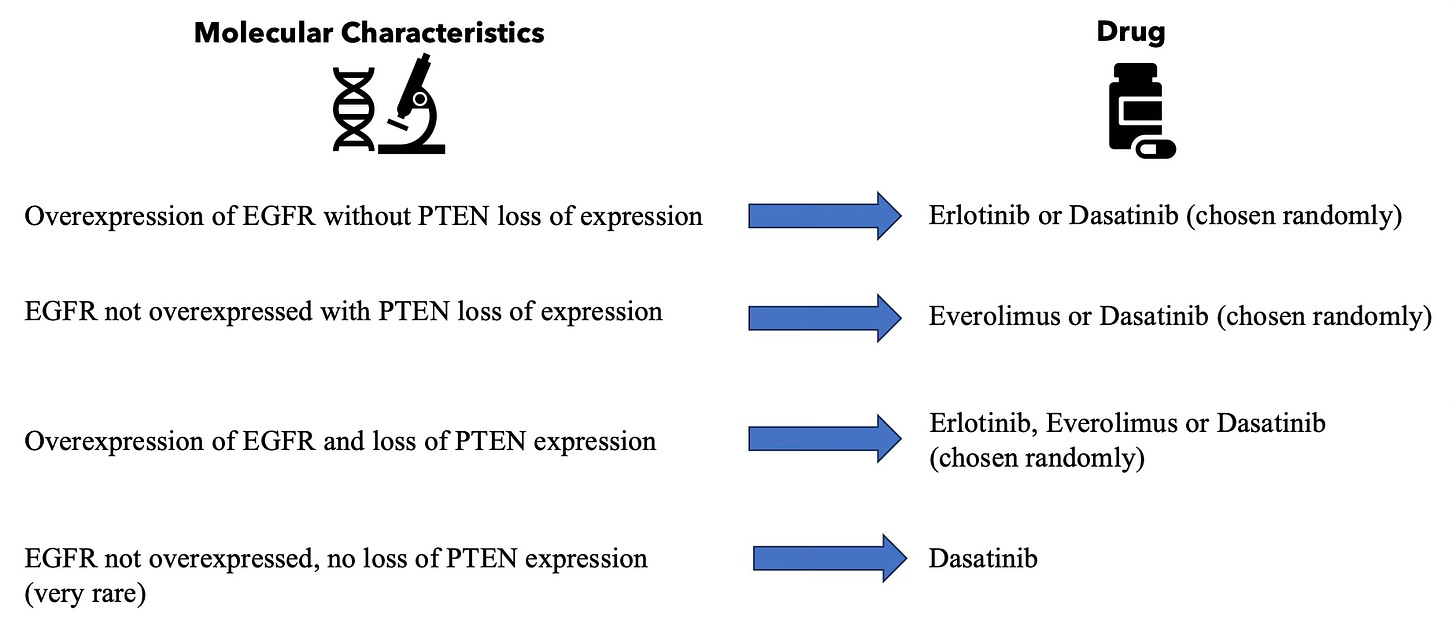
This approach is important because it tries to match the treatment to the tumour's specific characteristics, which could lead to better outcomes for patients. It's a way of tailoring cancer treatment to attack the tumour’s unique weaknesses.
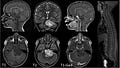
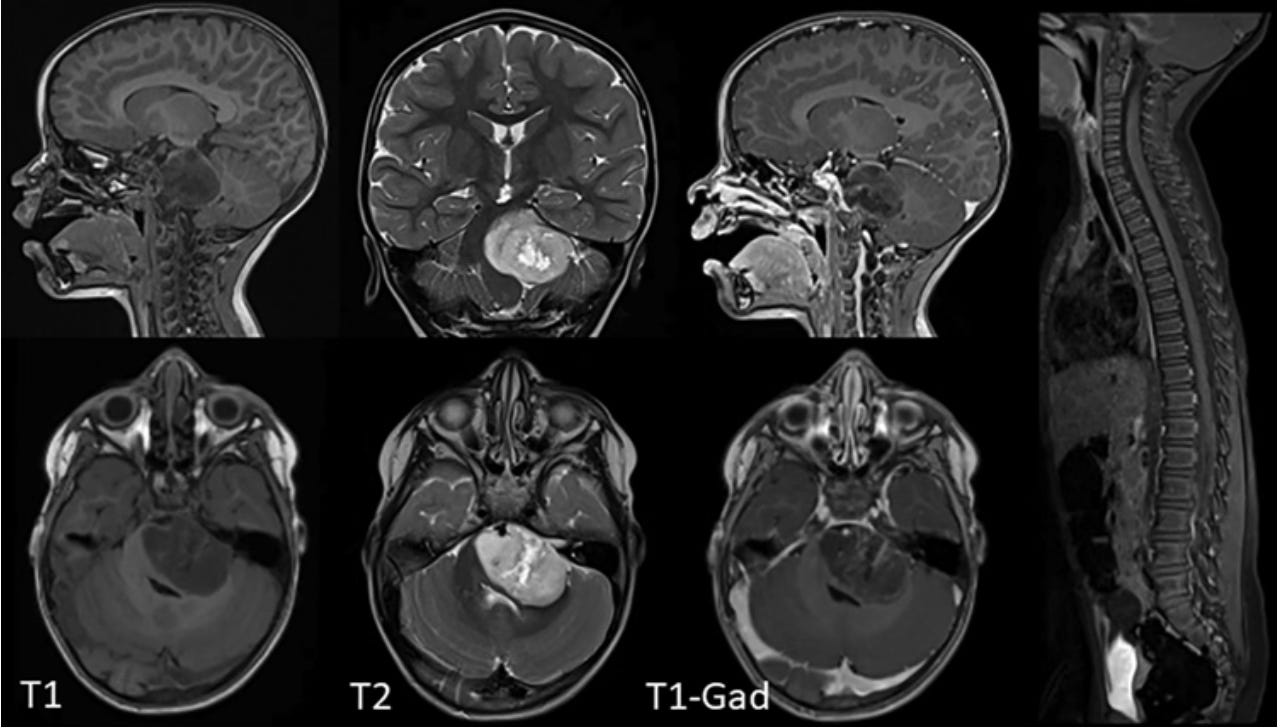
The miracle boy - Lucas’ story
Lucas was diagnosed with DIPG at six years old. Incredibly, against all odds, Lucas, now thirteen, shows no signs of the tumour. He's considered the first child to be ‘cured’ of brainstem glioma. While only time will tell if that is truly the case, what we have seen is a remarkable response that brings hope to so many families across the world faced with a similar diagnosis.

In desperation to seek alternative options, Lucas's family made the decision to travel from Belgium to France so he could participate in the BIOMEDE trial. Lucas responded remarkably well to the drug Everolimus, an mTOR inhibitor. The mTOR pathway plays a crucial role in cell growth and survival. In DIPG, mTOR contributes to its aggressiveness, growth and treatment resistance. The drug was chosen for him by random assignment in the trial.
Over time and through several MRI scans, Lucas’ tumour is no longer visible on scans. Even more encouraging, was that after Lucas stopped taking the medication a year and a half prior, his recovery held and there was still no evidence of disease.
The study investigators have revealed that the exact reasons for Lucas's full recovery are still not entirely clear, but are most likely influenced by a rare mutation in Lucas’ tumour, making it more susceptible to everolimus. While seven other children in the trial also survived for years post-diagnosis, Lucas's case stands out because his tumour was completely eradicated.
What now?
Researchers are now focusing on the genetic characteristics of the tumours from the patients in the trial. They are cultivating tumour organoids (see graphic inset) in the lab to study and replicate the conditions that led to Lucas's recovery. The goal is to reproduce the genetic nuances in these cell models and determine if they can be targeted similarly with drugs.

Turning a discovery into an approved drug is a marathon, not a sprint, but the shifting DIPG treatment landscape is sparking warranted optimism. Thanks to generous funding and groundbreaking trials like BIOMEDE, we're finally seeing a light at the end of the tunnel.
We’ve seen false hope too many times now, all for the sake of clicks following sensational headlines. Irresponsible journalists who know nothing about the disease but are keen to chase a ‘good news story’, with little context or nuance… But this time things are different and the hype is justified.
There's a real sense of hope that a cure for DIPG, at least for some, might not be too far off.
Onwards and upwards? - BIOMEDE 2.0: A Phase III trial
Following promising results from the phase II trial, the BIOMEDE 2.0 study, a phase 3 clinical trial, began on September 29, 2022.
The Phase III trial focuses on evaluating the efficacy of ONC201 in comparison with Everolimus for treating DIPG. Like Everolimus, ONC201 has anti cancer action as an mTOR inhibitor, but also posesses unique mechanisms, such as reactivating tumour suppressor genes.
The estimated primary completion date for this study is September 2028, with the overall study expected to conclude in September 2031. For more detailed information, you can visit ClinicalTrials.gov here.
A final note (IMPORTANT!)
If the BIOMEDE trial has caught your interest but you're unsure where to find information on clinical trials for your specific tumour type, Virtualtrials.org is an excellent resource. This platform offers detailed information on clinical trials that can be filtered for each individual's distinct situation.